Home
Technology
Pipeline
Services
About
News
Contact
A long-acting asparaginase for the treatment of Acute Lymphoblastic Leukemia
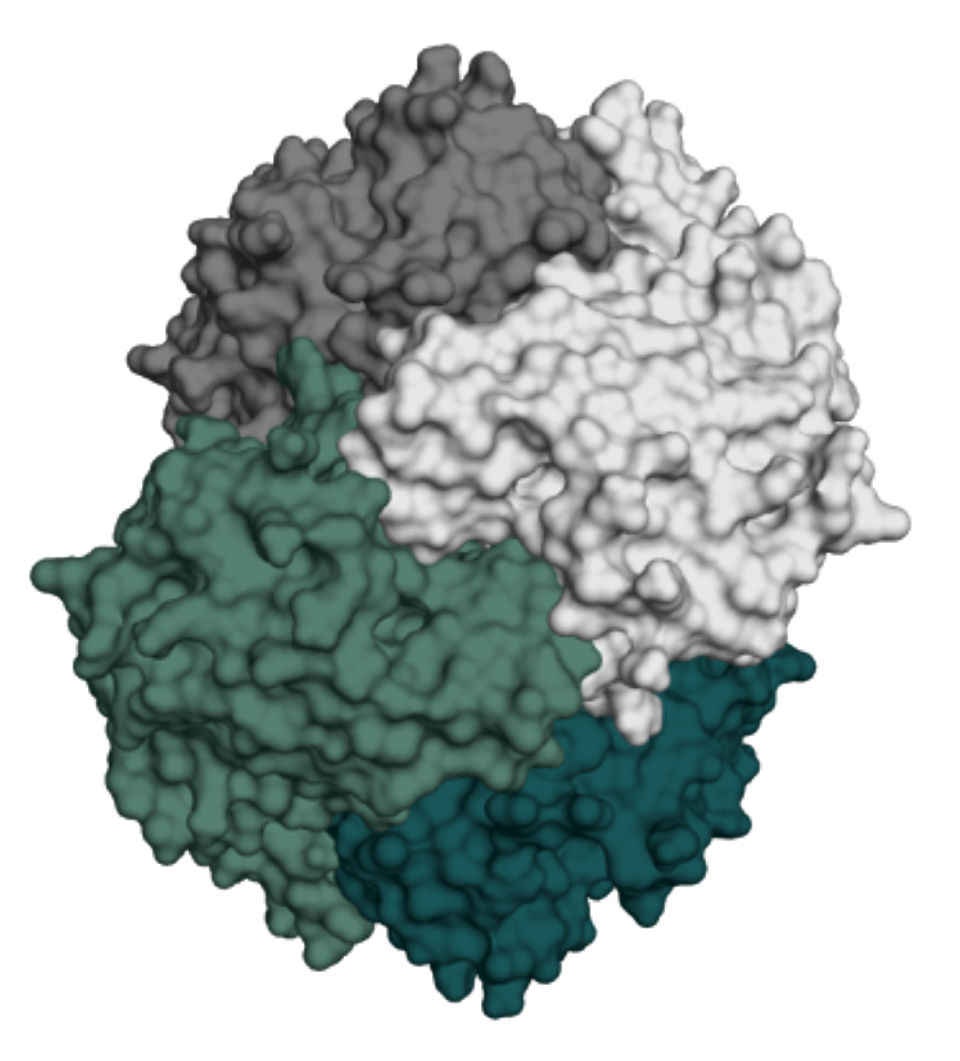
Acute lymphoblastic leukemia (ALL) is the most common childhood cancer. This hematologic malignancy affects both children and adults. Asparaginase is a key component of frontline treatment, yet current asparaginase-containing drugs face two major limitations:
Short half-life and frequent dosing
Hypersensitivity reactions
Incircular is developing a biobetter asparaginase candidate with best-in-class potential, stabilized using the proprietary INCYPRO platform.

Approach
INCYPRO reinforces the 3D structure of asparaginase, minimizing unfolding and degradation.

Design
Minimal sequence modification and unique structural design to preserve activity.

Solution
A long-acting asparaginase, with minimal hypersensitivity reactions and less frequent inpatient dosing, improving patients’ quality of life.
Development status and next steps