Home
Technology
Pipeline
Services
About
News
Contact
Pelay-Gimeno, M., Bange, T., Hennig, S., & Grossmann, T.N. Angew. Chem. Int. Ed. 2018, 57, 11164–11170. DOI: 10.1002/anie.201804506
This is the foundational publication establishing INCYPRO as a protein stabilization technology. It demonstrates how INCYPRO works: structure-based design, minimal protein modification and the resulting stability enhancements. The paper shows that the technology works across different proteins and protein folds.
This paper introduces the foundational In Situ Cyclization of Proteins (INCYPRO) technology. The approach uses C3-symmetric triselectrophilic crosslinkers that react with three aligned and surface-exposed cysteine residues that were introduced via site-directed mutagenesis. The result is a bicyclic protein topology that dramatically enhances structural integrity without altering the proteins core structure. Staphylococcus aureus Sortase A (SrtA), a transpeptidase widely used for protein labeling, was chosen as the model enzyme. SrtA variants (S1–S6) with cysteine pairs at different surface positions were designed and crosslinked, but showed only moderate increases in thermal stability. Introduction of three cysteines and crosslinking of those with triselectrophile t1 led to bicyclic variant S7-t1. In contrast to above monocyclic variants, S7-t1 drastically improved thermal stability. The INCYPRO protein displayed a melting temperature (Tm) of 70.6°C, representing a ΔTm of +11.2°C compared to wild-type SrtA (59.4°C). In addition, the bicyclic S7-t1 variant not only retained enzymatic transpeptidation activity at elevated temperatures (65°C) but also in the presence of the chaotropic denaturant guanidinium hydrochloride (GdnHCl). At 1M GdnHCl, S7-t1 maintained approximately 40% residual activity while wild-type SrtA was completely inactive. This demonstrated that INCYPRO-driven structural stabilization directly translates into functional resilience and can be utilized to widen the scope of protein applications. To demonstrate broader applicability, INCYPRO was applied to the KIX domain of the human CREB binding protein. Two crosslinked variants (K1-t1 and K1-t2) showed remarkable increases in melting temperature: ΔTm of +20.3°C and +24.3°C, respectively. This confirmed that the INCYPRO platform can be used to address and stabilize different protein families.
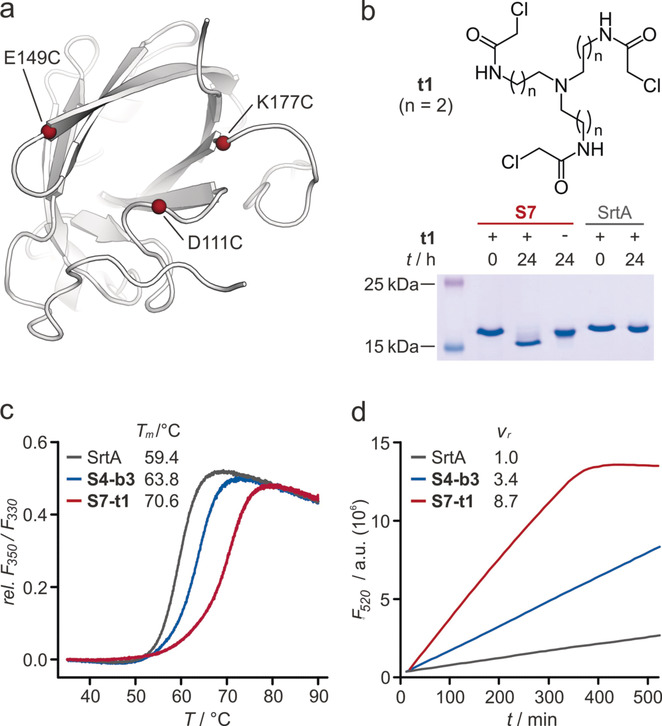
a) NMR structure of SrtA (PDB: 1ija) with positions of cysteine variations in S7 highlighted. b) Chemical structure of tris-electrophile t1 and Coomassie-stained SDS-PAGE gel showing protein bands after incubation with t1. c) Melting curves of SrtA, S4-b3, and S7-t1 including apparent Tm values. d) Fluorescence readout of enzymatic activity at 65 °C.